Eloira Hormone Intrauterine System Ius Levonorgestrel (Ing) IUD Birth Control Device
Price 2800.0 INR/ Unit
MOQ : 1 Piece
Eloira Hormone Intrauterine System Ius Levonorgestrel (Ing) IUD Birth Control Device Specification
- Wall Mounted
- No
- Portable
- Yes
- Condition
- NEW
- Material
- PLASTIC
- Feature
- Good Quality
- Storage Instructions
- Dry Place
- Use
- HOSPITAL
- Sterilized
- Yes
- Surface Finish
- Coating
Eloira Hormone Intrauterine System Ius Levonorgestrel (Ing) IUD Birth Control Device Trade Information
- Minimum Order Quantity
- 1 Piece
- Payment Terms
- Paypal, Telegraphic Transfer (T/T), Western Union
- Supply Ability
- 10 Pieces Per Day
- Delivery Time
- 3 Days
- Main Export Market(s)
- Asia, Australia, Central America
- Main Domestic Market
- All India
- Certifications
- ce
About Eloira Hormone Intrauterine System Ius Levonorgestrel (Ing) IUD Birth Control Device
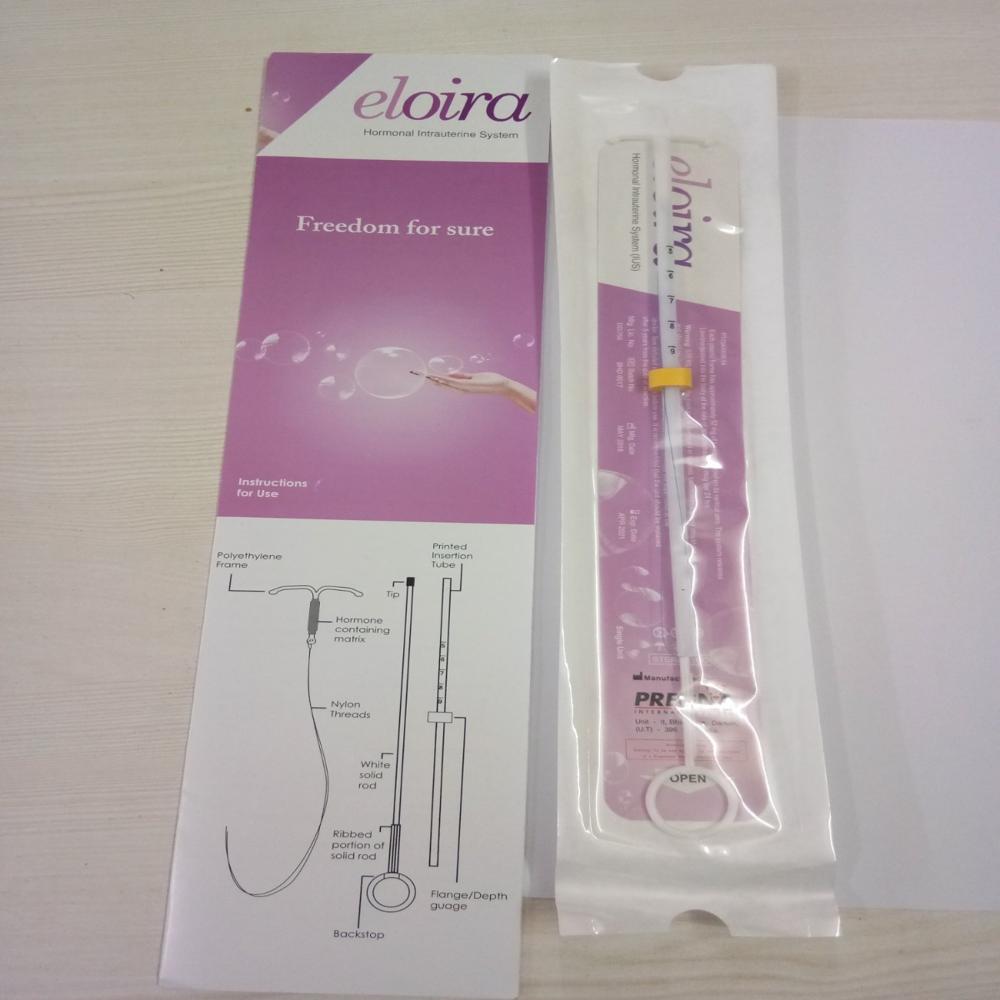
Intrauterine System (IUS)consists of a Plastic frame with a reservoir containing drug Levonorgestrel. Levonorgestrel(LNG)hormone is also found in other oral contraceptives. This drug reservoir releases a low daily a microdose of(LNG)into the uterine cavity.The cylinder/reservoir of the device contains hormone and is provided with a rate controlling membrane tubing that regulates the release of Levonorgestrel-LNG. TheLevonorgestrel IUDreleases the hormone at an approximate rate of 20 micrograms per day and effective for five years.IUSis a reversiblecontraceptivedevice & has high success rate and several non-contraceptivebenefits too.Thisintrauterine contraceptivedevice is embedded into the uterus by skilled provider. Thiscontraceptivedevice is thought to be a standout amongst the best forms ofbirth control deviceavailable today.FAQs of Eloira Hormone Intrauterine System Ius Levonorgestrel (Ing) IUD Birth Control Device:
Q: Is Eloira Hormone Intrauterine System sterilized for use?
A: Yes, the Eloira Hormone Intrauterine System is sterilized for safe use.Q: What material is used in the manufacturing of the Eloira Hormone IUD Birth Control Device?
A: The device is made of PVC and plastic materials.Q: Can the Eloira Hormone Intrauterine System be wall mounted?
A: No, this is a portable device and cannot be wall mounted.Q: Where should the Eloira device be stored?
A: It should be stored in a dry place.Q: Is the surface finish of the device coated?
A: Yes, the device has a coated surface finish.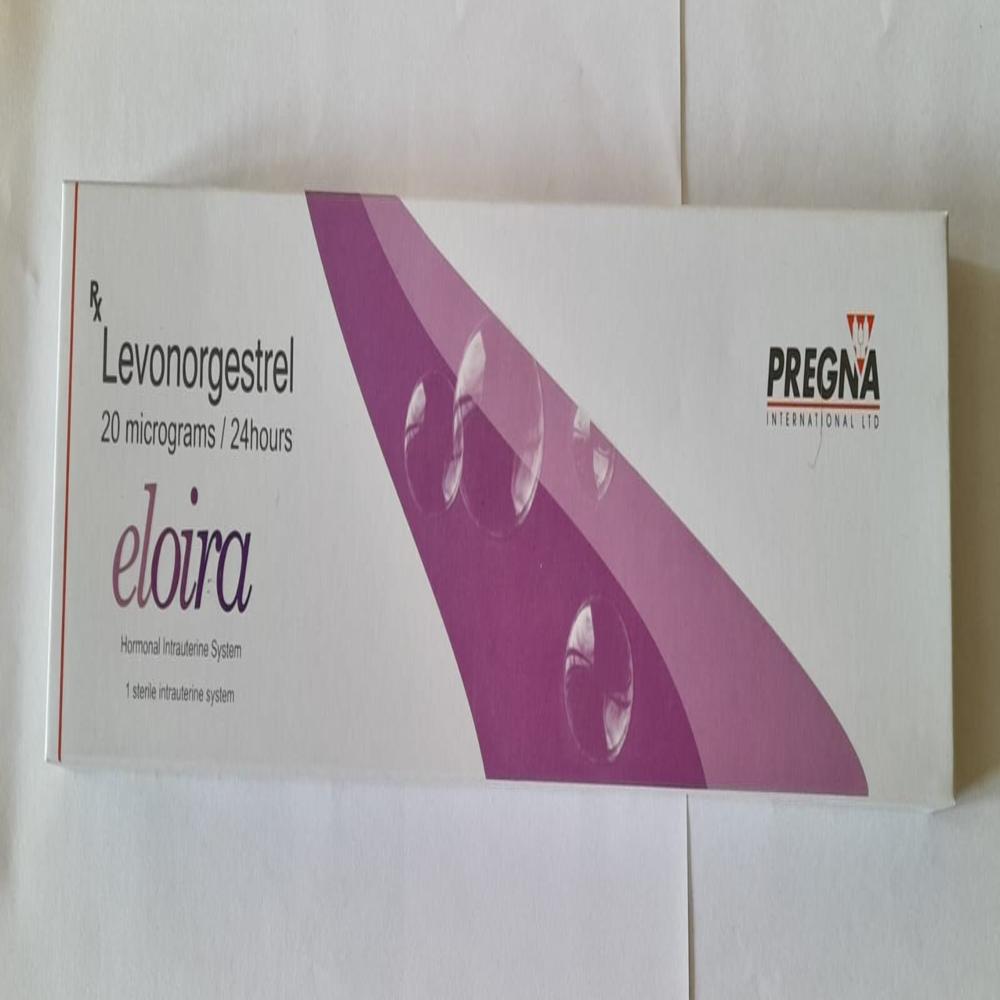
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Gynecology Instruments Category
Plastic HSG Procedure Kit With Flexible HSG Catheter HSG Procedure Tray With Shapeable HSG Catheter
Price 1500.0 INR / Piece
Minimum Order Quantity : 5 Pieces
Material : Plastic
Disposable : Yes
Equipment Type : Gynecology Instrument
Recyclable : Yes
Silicone Ring Pessary Silicone Vaginal Ring Pessary silicone tubal ring
Price 650.0 INR / Piece
Minimum Order Quantity : 5 Pieces
Material : PVC
Disposable : Yes
Equipment Type : Gynecology Instrument
Recyclable : Yes
Pap Smear kit Clear Sure Papsmear Test Kit One Wooden Spatula one Glass Slide Cervical Brush Swab Stick Sterile Set-4 Pc Sterilized
Price 12.0 INR / Piece
Minimum Order Quantity : 50 Pieces
Material : Plastic wooden
Disposable : Yes
Equipment Type : Gynecology Instrument
Recyclable : Yes
MVA Kits
Price 1600.0 INR / Unit
Minimum Order Quantity : 1 Piece
Material : Plastic
Disposable : No
Equipment Type : Gynecology Instrument

 Send Inquiry
Send Inquiry




 Send Inquiry
Send Inquiry Send SMS
Send SMS